Product Description
Buy Humatrope pen in US bodybuilding shop with worldwide delivery. We accept credit cards: MasterCard, Visa, AmEx and PayPal payments.
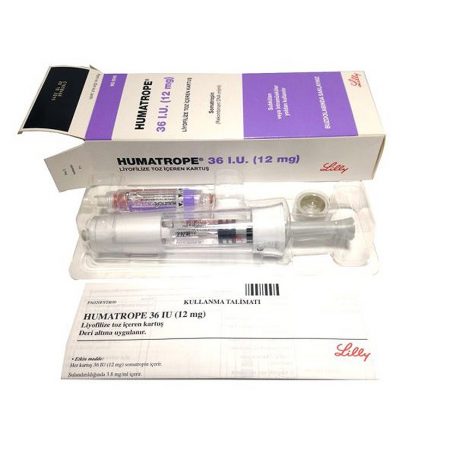
Our steroid shop offers high quality Human Growth Hormone for sale online. Eli Lilly’s Humatrope 36 I.U. 12mg is top quality Human Growth Hormone 36IU pen (Humatrope pen) from famous Hormones & Peptides producer.
Our online pharmacy delivers Hormones & Peptides worldwide, so we can ship your order to you fast, safe and discreet no matter where you live: USA, Canada, UK, Ireland, Spain, Germany, France, Italia, Holland, South Africa, Denmark, Sweden, Finland, Norway, Japan, New Zealand or other. We offer reshipping policy for missing, seized or damaged parcels.
We are working as steroid suppliers since March 2009 and have great experience in providing steroids for sale with complete comfort for customers.
Our friendly staff is always ready to help, all packages have tracking codes. We send notification emails for your order’s status change and you can always check current status via our special online service.
Humatrope 36 I.U. 12mg is widely known as Humatrope pen, Somatropin
Other alternative names for Humatrope 36 I.U. 12mg (Human Growth Hormone): Eli Lilly, Human Growth Hormone, HGH, Human Growth Hormone, HGH, Humatrope pen 36IU, Humatrope 36 I.U. 12mg 36IU.
All of these are different brands of the same active substance – Human Growth Hormone
Humatrope (Somatropin [rDNA origin] for injection) is used to treat:
- Children who do not make enough growth hormone on their own, have short stature associated with Turner syndrome, or have SHOX deficiency; have idiopathic short stature, which means they are shorter than 98.8% of other children of the same age and sex, are growing at a rate not likely to allow them to reach normal adult height, and for whom no other cause of short stature can be found; were born smaller than normal for the number of weeks of pregnancy and who do not catch up in height by 2 to 4 years of age
- Adults who have growth hormone deficiency that began either in adulthood (as a result of pituitary disease, hypothalamic disease, surgery, radiation therapy, or trauma) or in childhood. Patients treated for growth hormone deficiency in childhood whose bones have stopped growing should be reevaluated to determine if they should continue growth hormone
Important Safety Information for Humatrope
WHO SHOULD NOT TAKE HUMATROPE?
- You should not take Humatrope if you are having serious complications after undergoing open heart surgery, abdominal surgery, serious injuries involving many body systems, or life-threatening breathing problems. Deaths have been reported in such cases.
- Growth hormone should not be used in children with Prader-Willi syndrome who are severely obese or have a history of severe breathing problems. Deaths have been reported in children with Prader-Willi syndrome who are severely obese, have a history of blocked upper airways, sleep apnea (pauses in breathing during sleeping), or other severe breathing problems. Humatrope has not been approved for the treatment of patients with Prader-Willi syndrome.
- Humatrope should not be used if you have active cancer. Because growth hormone deficiency can be an early sign of some tumors in the brain or pituitary gland, the presence of these types of tumors should be ruled out by your doctor before you start Humatrope.
- Humatrope should not be used in children whose growth plates in their bones have closed.
- Humatrope should not be used in adults or children with diabetic retinopathy.
- If you are allergic to metacresol or glycerin, you should not mix Humatrope with the supplied diluent. Reactions at the injection site are the most common allergic reactions. If you have had an allergic reaction to growth hormone, you should not take Humatrope.
WHAT SHOULD I TELL MY DOCTOR BEFORE TAKING HUMATROPE?
- Tell your doctor if you take cyclosporine, hormone replacement therapy, insulin or other diabetes medications, drugs containing steroids, or drugs for seizures. These medications may need to be adjusted while taking Humatrope.
- There have been reports of death in children with Prader-Willi syndrome who recently started growth hormone therapy and had one or more of the following risk factors: obesity, history of severe breathing problems or sleep apnea (pauses in breathing during sleeping), or unidentified respiratory infection. Boys with one or more of these risk factors may be at greater risk than girls. A child with Prader-Willi syndrome should be evaluated by a doctor for signs of severe breathing problems or sleep apnea before starting growth hormone therapy.
- If you have pre-existing tumors, or growth hormone deficiency caused by abnormalities in the brain, you should be watched closely for worsening or returning of this condition. If you had cancer as a child and received radiation treatment to your head, you may have a greater risk of developing a new tumor if you are treated with growth hormone. The most common type of tumor reported in people treated with growth hormone after radiation to the head was a tumor of the covering of the brain, called a meningioma. If you developed growth hormone deficiency because you had a tumor inside the head or received radiation to the head, you should be monitored closely by your doctor during treatment with growth hormone. Children who have some rare genetic conditions may have a greater risk of developing a tumor if treated with growth hormone. If you have moles on your skin you should have these checked carefully for any changes while being treated with growth hormone.
- Your doctor should check your blood sugar regularly while you are taking Humatrope, especially if you have risk factors for diabetes. If you have diabetes or impaired glucose tolerance, your doctor should monitor your blood sugar closely during Humatrope therapy. New cases of type 2 diabetes have been reported in patients taking Humatrope.
- Cases of pancreatitis (inflammation of the pancreas) have been reported rarely in children and adults receiving growth hormone. Consult a doctor if you develop abdominal pain while taking Humatrope.
- Tell your doctor if you have any visual changes accompanied by headache, nausea, and/or vomiting while taking Humatrope. This may be a sign of increased pressure in the brain.
- Adults may retain water during therapy with Humatrope. This may be brief but may increase with higher doses of Humatrope.
- If you have hypopituitarism and are on standard hormone replacement therapy, your doctor should monitor your hormone replacement therapy closely while taking Humatrope.
- You should have your thyroid function tested periodically during Humatrope therapy. Thyroid hormone treatment may need to be started or adjusted.
- Slipped capital femoral epiphysis (fracture in the ball of the hip joint) can occur in children who have endocrine problems and in children who have rapid growth. Any child taking Humatrope who develops a limp or complains of hip or knee pain should be seen by a doctor to check for this.
- Progression of scoliosis (curvature of the spine) can occur in children who have rapid growth. Humatrope has not been shown to increase the occurrence of this condition.
- Children with Turner syndrome should be monitored closely by a doctor for ear infections and cardiovascular problems while taking Humatrope.
- You should rotate your injection sites to avoid breakdown of skin and fat. Seek prompt medical attention for any allergic reaction you experience to the injection of Humatrope.
- If you are pregnant or nursing, you should talk to your doctor before using Humatrope. It is not known whether Humatrope is excreted in human milk. Humatrope should be used during pregnancy only when clearly needed.
- Clinical studies in patients over 65 years of age have not been done. Elderly patients may be more sensitive to Humatrope and may experience more side effects.
- Because Humatrope may affect how two hormones, cortisol and cortisone, are processed in the body, people may discover they have hypoadrenalism after starting Humatrope therapy. In these cases, glucocorticoid replacement therapy would need to be started.
WHAT ARE THE POSSIBLE SIDE EFFECTS OF HUMATROPE?
- Common side effects reported in adults and children taking Humatrope include injection site reactions, allergic reactions to the diluent, and hypothyroidism. Additional common side effects in adults include swelling, joint pain, muscle pain, carpal tunnel syndrome, unusual skin sensations, and high blood sugar.
HOW SHOULD I STORE HUMATROPE?
- Humatrope must be kept refrigerated (36° to 46°F [2° to 8°C]) before and after it is mixed. Do not freeze. Once Humatrope has been mixed and is in liquid form, cartridges must be used within 28 days and vials must be used within 14 days. Throw away any unused Humatrope in a cartridge after 28 days and in a vial after 14 days. Before giving an injection, check the manufacturer’s expiration date on the cartridge or vial. Do not use the cartridge or vial if it has expired.
Buy Humatrope 36 I. U. 12mg online in Canada (Ihmisen Kasvuhormoni 36IU kynä). Los esteroides anabólicos en Estados Unidos: Humatrope 36 I. U. 12mg (de Menselijke Groei Hormoon 36IU pen). Achat de Humatrope 36 I. U. 12mg au Canada (Hormona de Crecimiento Humano 36IU pen)